Acid Oceans? & Oyster Shells
What’s Pure?
Visitor publish by Jim Steele
Printed July 14, 2020 within the Pacifica Tribune
(I wrote a white paper for the CO2 Coalition, offering extra particulars and references to look reviewed science regards how marine life counteracts ocean acidification. That paper will be downloaded right here )
Search the web for “acid oceans” and also you’ll discover hundreds of thousands of articles suggesting the oceans have gotten extra corrosive due the burning of fossil fuels, and “acid oceans” are threatening marine life. Though local weather modelers continually declare the oceans’ floor pH has dropped for the reason that 1800s, that change was by no means measured, because the idea of pH was not created till the early 1900s by beer-makers.
In 2003 Stanford’s Dr. Ken Caldeira coined the time period “ocean acidification” to generate public concern about growing CO2 . As New Yorker journalist Elizabeth Kolbert reported, “Caldeira advised me that he had chosen the time period ‘ocean acidification’ fairly intentionally for its shock worth. Seawater is of course alkaline, with a pH starting from 7.eight to eight.5—a pH of seven is impartial—which signifies that, for now, no less than, the oceans are nonetheless a good distance from truly turning acidic.” Nonetheless Caldeira’s time period “ocean acidification” evoked such undue fears and misunderstandings, we’re continually bombarded with catastrophic media hype and misdiagnosed causes of pure change.
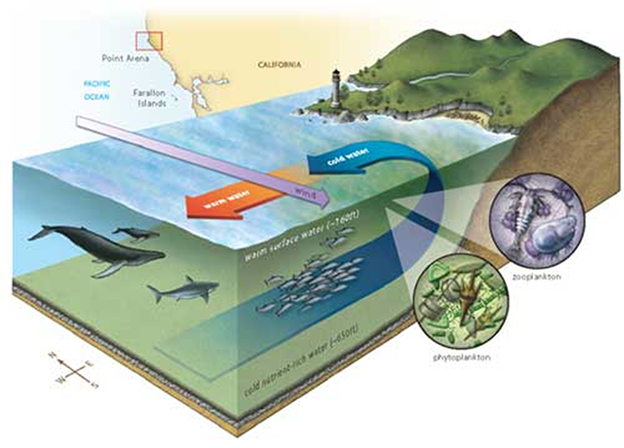
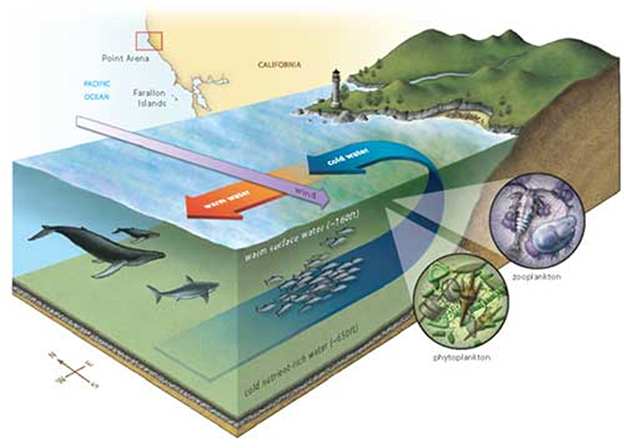
For instance, for almost a decade the media has hyped the 2006-2008 die-off of larval oysters in hatcheries alongside Washington and Oregon. They known as it a disaster attributable to rising atmospheric CO2 and the one answer was to cease burning fossil fuels. However it was an understanding of pure pH adjustments that offered the right options. Subsurface waters at a number of hundred meters depth naturally include larger concentrations CO2 and vitamins and a decrease pH than floor waters. Adjustments within the winds and currents periodically deliver these waters to the floor in a course of known as upwelling. Upwelling promotes a burst of life but additionally lowers the floor water pH. Not totally conscious of all of the CO2 dynamics, the hatcheries had made three errors.
First, they failed to acknowledge not all oyster species are effectively tailored to the low pH of upwelled water. The larvae of native Olympia oysters naturally survive intense upwelling occasions alongside the Washington coast as a result of that species “broods” its larvae. The larvae provoke their shells protected inside their dad and mom’ shells the place pH is extra managed. Nonetheless, the Olympia oysters had been over-harvested into close to extinction within the 1800’s.
So, fishermen imported the Japanese oyster, which is now the mainstay of the Washington and Oregon fisheries. Japanese oysters didn’t evolve inside an intense upwelling surroundings just like Washington’s coast. Every Japanese oyster merely releases over 50 million eggs into the water anticipating their larvae to outlive any gentle adjustments in pH throughout preliminary shell formation. Hatcheries didn’t understand the Japanese oyster’s larvae had a 6-hour window throughout which the larvae’s preliminary shell growth and survival was susceptible to low pH.
Second, as a result of cooler waters inhibit untimely spawning, hatcheries pumped cool water from the estuary within the early morning. As measured in coral reefs, photosynthesis raises pH in the course of the day, however nighttime respiration drops pH considerably. By pumping early morning water into their tanks, they imported estuary water at its lowest day by day pH. Lastly, they failed to acknowledge pure upwelling occasions transport deeper waters with naturally low pH into the estuary, additional reducing the pH of water pumped into their tanks.
Now, hatcheries merely pump water from the estuary later within the day after photosynthesis has raised pH. Scientists additionally developed a metering machine that detects intrusions of low pH waters, so hatcheries keep away from pumping water throughout upwelling occasions. As for many shellfish, as soon as the shell is initiated, a protecting layer prevents any shell corrosion from low pH situations. Drawback simply solved and disaster averted!
The simplistic concept that burning fossil fuels is inflicting the floor ocean to turn into extra acidic is predicated on the truth that when CO2 interacts with water a collection of chemical adjustments leads to the manufacturing of extra hydrogen ions which lowers pH. Sadly, all catastrophic analyses cease there. However dwelling organisms then reverse these reactions. Whether or not CO2 enters the floor waters through the environment or from upwelling, it’s shortly utilized by photosynthesizing plankton which counteracts any “acidification”. A share of the natural matter created within the sunlit waters sinks or is actively transported to depths, additional counteracting any floor “acidification’. Some natural matter sinks so quickly, CO2 is trapped at depths for a whole lot and hundreds of years. The dynamics that carry carbon to ocean depths largely explains why the oceans maintain 50 instances extra CO2 than the environment.
To take care of marine meals webs, it’s important that upwelling deliver sunken vitamins again into the daylight to allow photosynthesis. Upwelling additionally brings saved CO2 and low pH water to the floor. Wherever upwelling recycles vitamins and lowers floor pH, the best abundance and variety of marine life is generated.
Jim Steele is director emeritus of the Sierra Nevada Subject Campus, SFSU and authored Landscapes and Cycles: An Environmentalist’s Journey to Local weather Skepticism.
Like this:
Loading…